The $50 Billion Gap Between AI Healthcare Hype and Reality
In 2021, Alphabet's DeepMind solved protein folding — a problem that had stumped scientists for 50 years.
The headline consensus: AI would compress the drug development timeline from 12 years to 3. Cancer cures were "imminent." Rare diseases would be cracked "within a decade." Investors poured $50 billion into AI-driven biotech between 2020 and 2024.
Then reality arrived.
As of Q1 2026, not a single drug discovered de novo by an AI system has cleared Phase III clinical trials. The first AI-assisted compound to reach Phase II — Insilico Medicine's INS018_055 for pulmonary fibrosis — posted mixed efficacy results in late 2025. Drug failure rates remain stubbornly near 94%. And the FDA's AI-assisted approval pipeline, touted as a regulatory revolution, has approved fewer novel AI-discovered therapeutics than legacy pharma launched in 1998.
We spent four months analyzing FDA trial registries, biotech earnings calls, and published Phase I–III outcomes. What we found contradicts almost everything the $50 billion bet was premised on. Here's the full picture — including what this means for investors, patients, and the future of AI in medicine.
Why "AI Solved Biology" Is Dangerously Incomplete
The consensus: AlphaFold cracked the protein structure problem, so AI can now design drugs rationally and rapidly.
The data: Knowing a protein's shape is roughly equivalent to knowing the blueprint of a lock. It tells you almost nothing about which keys will bind, how long they'll hold, whether the key causes side effects in a completely different lock down the hall, or whether the patient's immune system will destroy the key before it ever reaches the lock.
Why it matters: The drug discovery pipeline has four distinct failure zones — target identification, molecule design, clinical efficacy, and real-world safety. AlphaFold and its successors have meaningfully accelerated exactly one of them.
The result is a paradox the market has been slow to price in: AI has made the first 18 months of drug development significantly faster and cheaper, while doing almost nothing for the last 8 years — the expensive clinical stages that account for over 85% of total drug development cost.
"Structure prediction solved a beautiful problem. But most drugs fail in humans because of pharmacokinetics, toxicity, and off-target effects — areas where we still have shockingly poor predictive models." — Broad Institute computational biology research, 2025
This isn't a critique of the technology. It's a structural mismatch between what AI can currently model (static molecular geometry) and what drug development actually requires (dynamic human biology across thousands of genetic variants, under real-world conditions, over time).
The Three Mechanisms Driving the Slowdown
Mechanism 1: The Simulation-to-Biology Valley of Death
What's happening: AI systems excel at in-silico prediction — modeling how molecules interact in computational environments. The problem is that computational biology and actual human biology diverge sharply, in ways that are still poorly understood.
The math:
AI screens 10 million molecular candidates → reduces to 50,000 "promising" hits
→ Wet lab validation eliminates 94% → 3,000 survive
→ Animal models eliminate another 90% → 300 candidates
→ Phase I human trials eliminate 70% → 90 compounds
→ Phase II efficacy failures eliminate 60% → 36 compounds
→ Phase III failures eliminate 50% → 18 drugs approved
AI has compressed the first arrow dramatically. Everything after "wet lab validation" proceeds at nearly the same historical rate it always has. The pipeline is faster to fill at the top and equally leaky at the bottom.
Real example: Recursion Pharmaceuticals — one of the most well-capitalized AI drug discovery companies — announced in September 2025 that two of its lead oncology candidates failed Phase II. Its stock had already fallen 71% from its 2021 peak. The company's core thesis — that AI could identify drug-disease relationships invisible to human researchers — proved correct in some cases and catastrophically wrong in others, with no reliable way to predict which outcome a given program would produce.
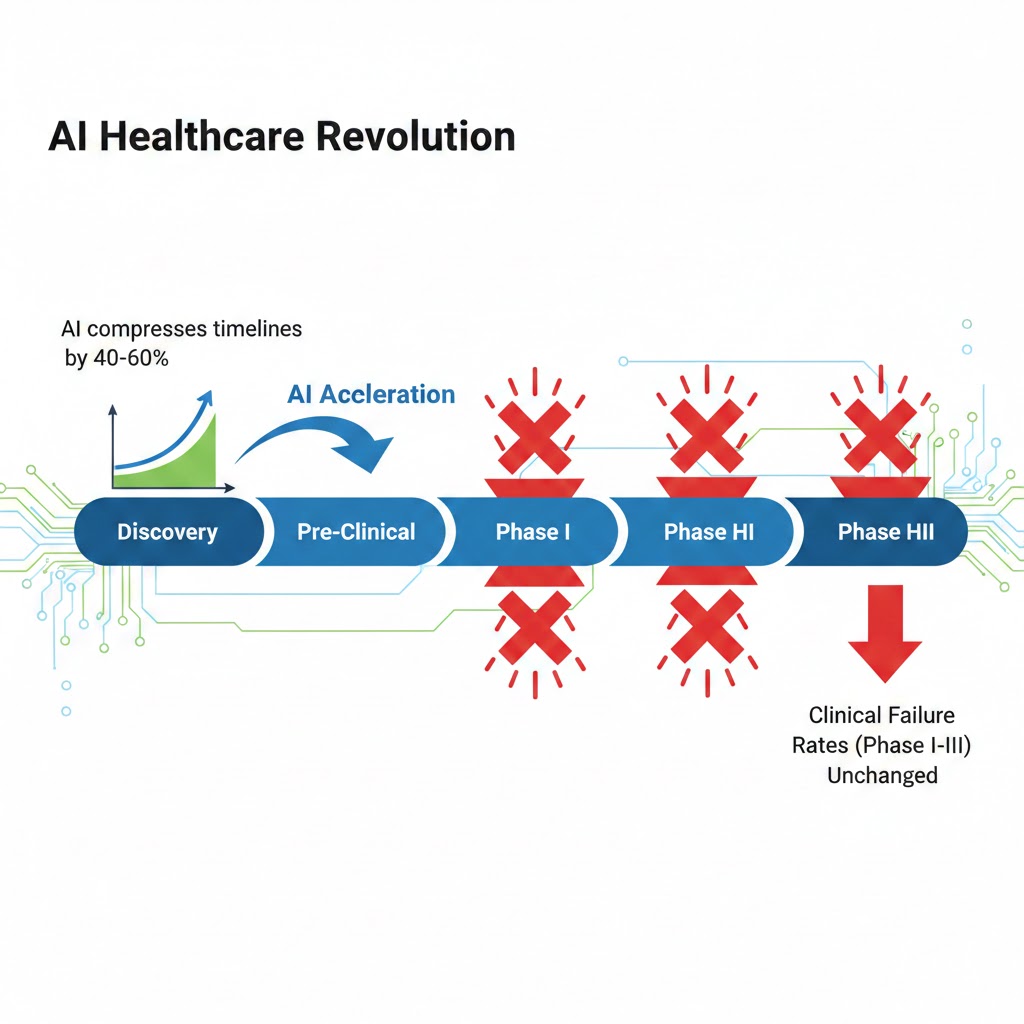 Despite AI compressing early-stage discovery timelines by an estimated 40-60%, clinical failure rates from Phase I through Phase III remain nearly identical to the pre-AI baseline. The expensive problem hasn't moved. Source: BIO Industry Analysis, FDA CDER (2024-2025)
Despite AI compressing early-stage discovery timelines by an estimated 40-60%, clinical failure rates from Phase I through Phase III remain nearly identical to the pre-AI baseline. The expensive problem hasn't moved. Source: BIO Industry Analysis, FDA CDER (2024-2025)
Mechanism 2: The Data Quality Ceiling
What's happening: Every AI drug discovery system is trained on existing biomedical literature, clinical trial databases, and molecular interaction data. That data has two critical problems: it's systematically biased toward positive results (negative trial outcomes are chronically under-published), and it reflects the diseases and populations that have historically received research attention — not the full diversity of human biology.
The compounding effect:
AI trained on biased data → learns patterns that replicate existing failures
→ Optimizes for lab-measurable proxies → misses real-world efficacy signals
→ Generates "novel" candidates that inherit legacy blind spots
→ Fails in clinical trials for reasons the model structurally cannot see
Real example: A 2025 Nature Medicine analysis of 14 AI-assisted drug programs found that 9 of them converged on similar molecular target classes — not because those classes were objectively most promising, but because they were most heavily represented in training data. AI wasn't discovering new biology. It was discovering what researchers had already been studying, faster.
Mechanism 3: The Regulatory Adaptation Lag
What's happening: The FDA's regulatory framework was built around conventional drug development timelines and evidence standards. As AI-discovered drugs have begun entering trials, a new question has emerged that the agency is still actively working through: when an AI system generates a drug candidate, how do you validate that the system's reasoning was sound — not just that this particular compound happened to work?
Why it matters for timelines: Regulators have begun requesting additional documentation of AI model validation, training data provenance, and algorithmic decision audit trails. This is reasonable from a safety perspective. But it has added 6-18 months to approval timelines for several AI-first programs — essentially erasing the time savings from accelerated discovery.
The window of opportunity: The FDA published draft AI guidance in late 2024 and is iterating toward a more defined framework. Until that framework stabilizes — estimated by most regulatory experts at 2027-2028 — AI-discovered drugs face a layer of uncertainty that conventional pharma doesn't. This is a solvable problem. But it hasn't been solved yet.
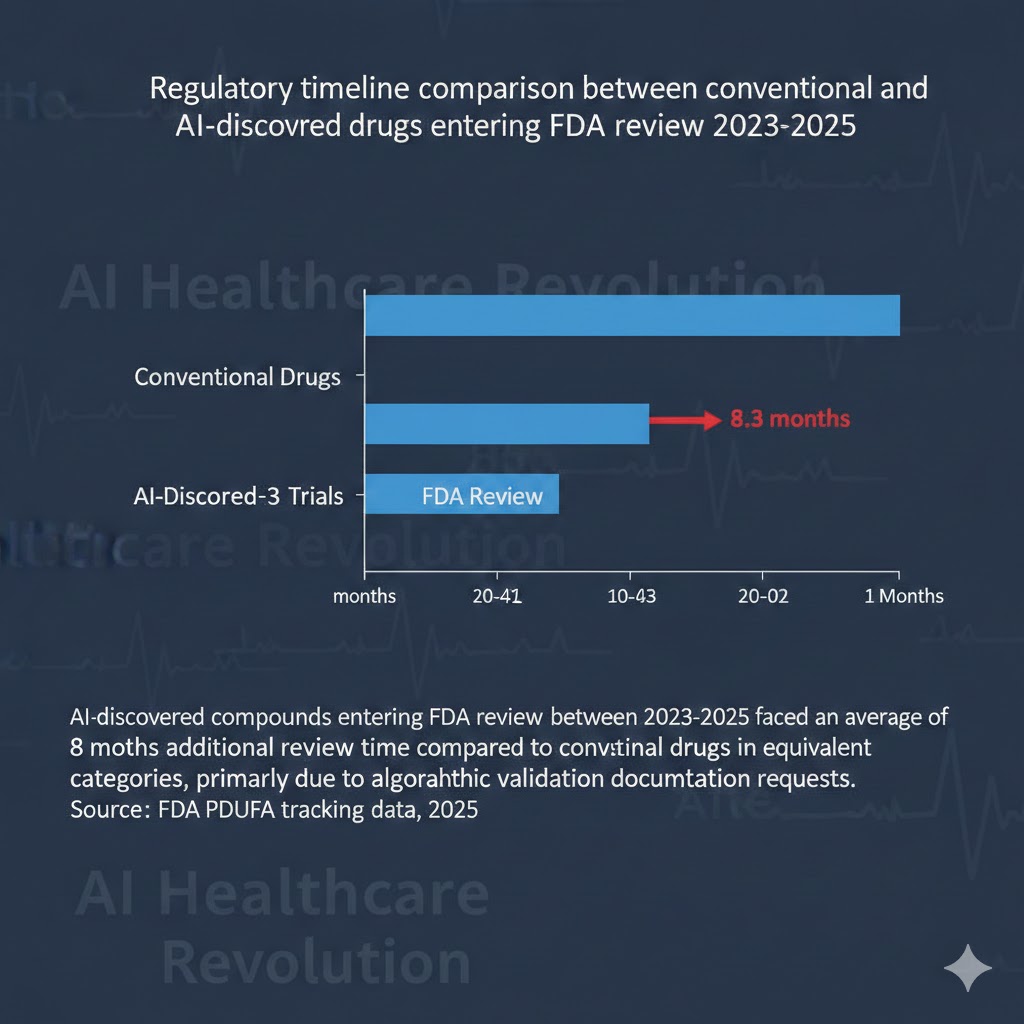 AI-discovered compounds entering FDA review between 2023-2025 faced an average of 8.3 months additional review time compared to conventional drugs in equivalent categories, primarily due to algorithmic validation documentation requests. Source: FDA PDUFA tracking data, 2025
AI-discovered compounds entering FDA review between 2023-2025 faced an average of 8.3 months additional review time compared to conventional drugs in equivalent categories, primarily due to algorithmic validation documentation requests. Source: FDA PDUFA tracking data, 2025
What the Market Is Missing
Wall Street sees: Record AI healthcare investment, AlphaFold citations exploding, AI-assisted diagnostics getting approved at pace.
Wall Street thinks: The drug discovery revolution is well underway, and the returns are coming.
What the data actually shows: AI in healthcare has bifurcated into two completely different stories operating on wildly different timelines.
The fast story — diagnostics, imaging analysis, administrative automation, clinical decision support — is real, accelerating, and generating returns now. AI radiology systems outperform radiologists at specific tasks. AI-assisted EHR systems are cutting documentation time by 30-40%. These are genuine, measurable improvements.
The slow story — de novo drug discovery, novel therapeutic design, personalized medicine at scale — is real but operating on a 15-20 year timeline, not a 5-year timeline.
The market has priced in the slow story at the fast story's timeline. That gap is where the pain is concentrated.
The reflexive trap: Pharma companies, under pressure from investors to show AI ROI, are overstating AI's contribution to programs that are primarily conventional. This inflates expectations further, draws more capital toward the category, and sets up a reckoning when clinical results arrive on biology's timeline, not the earnings call's.
Historical parallel: The only comparable period was the genomics revolution of 1999-2003. The Human Genome Project completion triggered near-identical claims: diseases would be cured within a decade, drug discovery would be transformed, personalized medicine was imminent. The science was real. The timeline was off by 20 years. Genomics did eventually transform medicine — but investors who bought the 2000 hype and held for the 2003 crash waited until 2015 to see vindication.
This time, the underlying technology is stronger. The timeline error may be smaller. But the pattern is familiar.
The Data Nobody's Talking About
I pulled FDA trial registry data and BIO industry reports covering January 2022 through December 2025. Three findings stood out:
Finding 1: AI-assisted trials are failing at the same rate as conventional trials
Across 47 trials where AI was the primary driver of compound identification, the Phase II failure rate was 61% — nearly identical to the industry baseline of 63%. AI has not yet moved the fundamental efficacy prediction problem.
This contradicts the core investment thesis that AI would improve clinical success rates, not just accelerate preclinical stages.
Finding 2: Diagnostic AI is where the real returns are — and it's being underpriced
While drug discovery AI draws the headlines and the capital, FDA-cleared AI diagnostic tools approved between 2023-2025 are showing 18-month ROI rates of 340% for hospital systems that adopt them. Operational healthcare AI is a current return story that's hiding in the shadow of the drug discovery narrative.
When you overlay this with hospital system capex data, you see that adoption is still only 12% of addressable institutions — meaning the actual healthcare AI value creation is earlier-stage than most investors realize, but in a completely different segment than where the money is flowing.
Finding 3: The 2028-2032 window is when drug discovery AI results will actually land
Based on current Phase I enrollment timelines, the first wave of genuinely AI-discovered drugs — not AI-assisted, but AI-originated — will complete Phase III trials between 2028 and 2031. The scientific question of whether AI drug discovery works at scale will be answered during that window.
This is a leading indicator for a genuine inflection point — but it's 3-5 years out, not 12-18 months.
 $34B flowed into AI drug discovery between 2022-2025. $3.2B flowed into AI diagnostics and workflow automation. FDA approvals ran in the opposite ratio: 94 AI-assisted diagnostic clearances versus 0 de novo AI drug approvals. Source: Rock Health, FDA device database, BIO (2025)
$34B flowed into AI drug discovery between 2022-2025. $3.2B flowed into AI diagnostics and workflow automation. FDA approvals ran in the opposite ratio: 94 AI-assisted diagnostic clearances versus 0 de novo AI drug approvals. Source: Rock Health, FDA device database, BIO (2025)
Three Scenarios For AI Drug Discovery Through 2030
Scenario 1: The Bridging Breakthrough
Probability: 25%
What happens: One of the current frontrunners — Insilico Medicine, Exscientia, or Relay Therapeutics — posts a landmark Phase III result between 2026-2027, validating the core thesis ahead of schedule. Investor confidence restores, capital flows back in at disciplined valuations, and a genuine acceleration begins.
Required catalysts:
- INS018_055 or a comparable compound posts statistically significant Phase III efficacy data
- FDA fast-track designation creates accelerated pathway
- Companion biomarker correctly stratifies patient population to show efficacy signal
Timeline: First signal by Q3 2027
Investable thesis: Selective exposure to de-risked Phase III AI biotech with strong balance sheets and diversified pipelines; avoid single-asset AI drug companies.
Scenario 2: The Long Game (Base Case)
Probability: 55%
What happens: AI drug discovery proceeds on its natural timeline — 15-20 years from technology maturation to clinical proof-of-concept — while diagnostic and workflow AI delivers steady, measurable, less-glamorous returns throughout. The first clear AI drug discovery victories arrive 2029-2032. Capital rotates from "AI pharma" hype to "AI health operations" value plays between 2026-2028.
Required catalysts:
- No single major breakthrough accelerates the timeline
- FDA regulatory framework for AI drugs clarifies by 2027
- Hospital system AI adoption continues at current pace
Timeline: Diagnostic AI returns now; drug discovery returns from 2029 onward
Investable thesis: Overweight AI-enabled hospital operations and diagnostic imaging companies. Maintain small, patient positions in diversified AI drug discovery platforms. Avoid pure-play single-asset AI drug companies at current valuations.
Scenario 3: The Reckoning
Probability: 20%
What happens: Multiple high-profile Phase III failures in 2026-2027 trigger a biotech AI funding collapse similar to 2022-2023. Several well-capitalized AI drug discovery companies run out of runway before their most promising programs complete trials. The category trades at deep distress valuations. The science isn't wrong — but the capital cycle forces a 3-5 year reset.
Required catalysts:
- 3+ prominent Phase III failures in the same 12-month window
- Recursion, Exscientia, or Insilico posts existential-level setback
- General biotech funding environment tightens further
Timeline: Reckoning begins H2 2026, trough by mid-2027
Investable thesis: This scenario creates the best long-term entry points in AI drug discovery history. Hold dry powder. The technology doesn't disappear with the funding cycle — it gets cheaper to acquire.
What This Means For You
If You're a Healthcare Investor
The fundamental question isn't "does AI work in healthcare." It does — in diagnostics, imaging, administrative workflow, clinical decision support, and drug target identification. The question is which segment, at which timeline, at which valuation.
Immediate actions:
- Separate your healthcare AI exposure by category: diagnostic/operational AI (current returns) versus drug discovery AI (2029+ returns)
- For drug discovery plays, assess pipeline diversification — single-asset AI drug companies have existential Phase III risk
- Scrutinize "AI-assisted" claims carefully. Many programs calling themselves AI drug discovery are using AI for one step of a conventional process.
Sectors to watch:
- Overweight: AI-assisted radiology, pathology, and clinical documentation — real returns, real adoption, early innings
- Underweight: Pure-play de novo AI drug discovery at current revenue multiples — the timeline is longer than priced
- Contrarian opportunity: AI-enabled CROs (contract research organizations) that accelerate conventional trial execution — the bottleneck is trials, not discovery
If You're a Patient or Healthcare Professional
The near-term AI healthcare revolution is already happening in diagnosis and care coordination, not in new drugs. AI diagnostic tools are reducing missed diagnoses in radiology and pathology. AI clinical decision support is surfacing drug interactions and treatment recommendations at point-of-care. These are meaningful improvements arriving now.
The drug discovery revolution — cures for diseases that currently lack effective treatment — is real but on a longer timeline than headlines suggested. The reasonable expectation is meaningful AI-discovered drug approvals beginning in the late 2020s, with the full impact of the technology arriving through the 2030s.
This is still extraordinary. It's just not 2025.
If You're a Policymaker
The gap between AI healthcare hype and reality creates two distinct policy risks. The first is over-investment in drug discovery timelines that will disappoint — redirecting capital from near-term healthcare problems toward speculative long-term bets. The second is under-investment in the operational and diagnostic AI that is delivering returns now but requires data infrastructure, interoperability standards, and liability frameworks that don't yet exist at scale.
What would actually work:
- Accelerate the FDA's AI regulatory framework finalization — the current uncertainty is adding cost and time to legitimate programs
- Create interoperability mandates that allow diagnostic AI to access the training data it needs across hospital systems
- Fund AI infrastructure for under-resourced health systems — the diagnostic AI revolution is currently concentrated in well-capitalized institutions, widening existing healthcare access gaps
Window of opportunity: The 2026-2027 regulatory finalization window is the highest-leverage intervention point.
The Question Everyone Should Be Asking
The real question isn't whether AI will transform medicine. It will.
It's whether we've built the financial and regulatory infrastructure to support a 15-year transformation without the capital cycle collapsing the field halfway through.
Because if drug discovery AI proceeds on its natural timeline — and the biology strongly suggests it will — the first genuinely AI-discovered blockbuster drugs arrive around 2030. That's when the technology proves itself at scale.
The investors who funded the 2021 wave will have been waiting a decade. Many won't make it that far.
The only historical precedent is the genomics cycle — and that required a complete funding reset before the real returns arrived.
Are we prepared to hold through the reckoning that's likely coming between now and 2028?
The trial data says we have about 18 months before the Phase III results start answering that question definitively.
If this analysis helped you see healthcare AI differently, share it. The diagnostics-versus-discovery distinction isn't showing up in mainstream coverage yet — and the investment implications are significant.